news

news
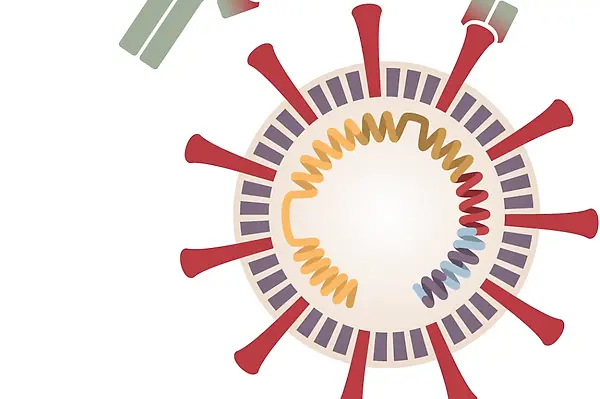
NCSSM Dean of Science Dr. Amy Sheck is participating as a test subject in the phase 3 clinical trial of a COVID-19 vaccine being developed by Pfizer and BioNTech, which uses messenger RNA to prompt the human body to produce an immune response to fight off the novel coronavirus. Earlier this month, Pfizer reported a preliminary finding from its clinical trials that its vaccine was about 90 percent effective at preventing people from contracting the disease. We asked Sheck about her experience of taking part in this historic clinical study.
Have you participated in drug or vaccine trials in the past?
This is my very first one.
How did you become aware of the opportunity to participate?
Through my family doctor, my general practitioner. He has run clinical trials before for other drugs. This one, I was really motivated to participate, so I volunteered. Both my husband and I are in the trial.
Why did you feel so highly motivated?
Obviously, there is a lot we can do as a society to bring the pandemic under control, but I think vaccines are the most important thing that needs to happen next. I’m a big fan of vaccines in general as one of the most important and effective public health measures next to handwashing. This vaccine cannot go forward into general use without 44,000 volunteers participating in the study, so being one of those is my little piece of helping that to happen.
What has participating in the trial required of you so far?
It’s been pretty easy. This started in August. The first step was informed consent. We met with the doctor, and there was a lot of information you have to read. The doctor is obliged to educate us about what we’re getting into, and then we had to agree. Then, I had to get tested for COVID. Once I knew I was negative, I had to get two injections three weeks apart. Of course, it’s blind — did I get the vaccine or the placebo? I don’t know.
I agreed to record my health status once a week, whether I have COVID symptoms or not. I’ll also come back several times over two years to get blood drawn; they are testing for antibodies. I should also be very upfront that trial participants are also paid for their time. For each visit I get paid about $100 or so; over two years that’s about $600. I think that’s important, because when you have a trial of 44,000 people, you need to get everyone (from different socioeconomic backgrounds) to participate, and paying people for their time helps make that possible.
One of the interesting criteria for participation was that you not be completely isolating at home. The trial is only useful if people have some exposure out in the world, because they compare the number of cases in people who got the vaccine with the cohort that got only the placebo to see if the vaccine made a difference. So the fact that I’m teaching on campus at NCSSM makes me a good vaccine trial candidate.
Do you have a preference or wish about whether you received vaccine vs. placebo? And when will you find out which you received?
Oh, I would much rather get the vaccine, for sure. I would be very happy if that were the case. I am not afraid; I’ve never really been afraid of vaccines and was not worried about possible side effects. I did have local swelling at the injection site. I was tired for about a day, and I think after the second dose I had some chills.
When Pfizer actually gets approval for this vaccine, likely mid-December, they will break the blind and tell people if they got the placebo. People who did will then be allowed to receive the vaccine as a benefit of participating in the trial. I’ll continue to participate in the trial for two years to see how long the antibodies last. (Editor’s note: Sheck later earned she indeed received the active vaccine during the trial.)
Surveys show that many Americans are nervous about receiving a vaccine that is proceeding much more quickly than usual through trials and regulatory hurdles. Your concerns are obviously low enough that you felt comfortable participating. What’s your assessment of the risk of a new vaccine versus the risk we all face already of contracting COVID-19?
To me, they don’t really compare. The risk of COVID is the risk of death, but even short of that, there’s lung damage, the neurological damage, and the effects that can carry on for months. That is much more concerning to me than any possible side effect of the vaccine. The most serious side effect is that the vaccine can make your immune system overreact to the pathogen.
Yes, normally developing this vaccine would take five years, and here we are doing it in 18 months, so this is jaw-dropping. Pfizer extended the trial to see more cases of COVID among test subjects to gain the confidence of the public. You know, 44,000 people is a really big data set, and the preliminary results look really strong to me. The results of the trial are fantastic, better than anyone really dreamed. The scientist in me would love to know more about the data set, the demographics and occupations of people participating, and the biology of why the vaccine did not work for some small percentage of people.
Are NCSSM students studying the vaccine development process and the pandemic?
Yes, there are many important and interesting angles on this pandemic, and we want to give our students the chance to study those. There is a lot of expertise among our faculty, as well. We offered a Computing COVID-19 short course over the summer looking at the evolution of the coronavirus. We also have an Open Enrollment course in global health, a residential course in immunology, and an online course in epidemiology. (Science instructor) Bob Gotwals and I are working on a virtual COVID-19 symposium to be held in March. We have invited some really high profile researchers, and we will announce details in the weeks to come.